Symbol
Title
Definition

Manufacturer
Indicates the medical device manufacturer.

Distributor
Indicates the entity distributing the medical device into the locale.

Importer
Indicates the entity importing the medical device into the locale.
.png?width=159&height=64&name=EU%20REP%20(2).png)
Indicates the Authorized Representative in the European Union.

Authorized Representative in the European Community
Indicates the Authorized Representative in the European Community.
*Note: This symbol was replaced in the amendment released for ISO 15223-1:2021/AMD 1:2025
.png?width=197&height=79&name=EU%20REP%20(1).png)
Indicates the authorized representative in the identified country or jurisdiction identified in the two-letter or three-letter country code on the label.

Authorized Representative in the Switzerland Community
Indicates the Authorized Representative in Switzerland.

Date of Manufacture
Symbol for date of manufacture. This symbol is accompanied by a date.
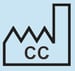
Country of Manufacture
To identify the country of manufacture of products.
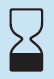
Use-by Date
Indicates the date after which the medical device is not to be used.

Lot Number
Indicates the manufacturer's lot number so that the lot can be identified NOTE: Synonyms for "Lot Number" are "barch code" and "batch number"

Reference Number
Indicates the manufacturer's reference number so that the medical device can be identified NOTE: Synonyms for "reference number" are "catalogue number" and "reorder number"

Unique Device Identifier
Indicates a carrier that contains unique device identifier information.

Sterilized Using Ethylene Oxide
Indicates a medical device that has been sterilized using ethylene oxide.

Do Not Resterilize
Indicates a medical device that is not to be resterilized.

Non-Sterile
Indicates a medical device that has not been subjected to a sterilization process.
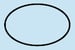
Single Sterile Barrier System
Indicates a single sterile barrier system.

Do Not Use if Package is Damaged
Indicates a medical device that should not be used if the package has been damaged or opened.

Do Not Reuse
Indicates a medical device that is intended for one use, or for use on a single patient during a single procedure.

Caution
Indicates that caution is necessary when operating the device or control close to where the symbol is placed, or that the current situation needs operator awareness or operator action in order to avoid undesirable consequences.

Consult Instructions For Use
Indicates the need for the user to consult the instructions for use.

Prescription Only
Caution: Federal law restricts this device to sale by or on the order of a physician.

CE Mark
CE mark Product conforms with the essential requirements in the MDR 2017/745.

CE Mark
CE mark including NSAI (notified body) identification number. Product conforms with the essential requirements in the MDR 2017/745 as well as in the European Medical Devices Directive 93/42/EEC.
![]()
Brightness
To identify the brightness control.
![]()
Type CF Equipment
To identify a type CF applied part complying with IEC 60601-1.
![]()
Type BF Equipment
To identify a type BF applied part complying with IEC 60601-1.
![]()
GS1 specific device identifier (Global Trade Item Number)
Indicates the device identifier portion of the Unique Device Identifier (UDI).

Medical Device
Indicates the product is a medical device.